R22 Suppliers, R22 Manufacturers.

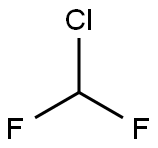 |
| Difluorochloromethane Chemical Properties |
| mp | -146°C |
| bp | -40.8°C |
| density | 1,18 g/cm3 |
| Water Solubility | Slightly soluble |
| Stability: | Stable. |
| CAS DataBase Reference | 75-45-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Methane, chlorodifluoro-(75-45-6) |
| EPA Substance Registry System | Methane, chlorodifluoro-(75-45-6) |
| Safety Information |
| Hazard Codes | F |
| Risk Statements | 40-59 |
| Safety Statements | 23-59 |
| RIDADR | 1018 |
| Hazard Note | Non-Flammable |
| HazardClass | 2.2 |
| Hazardous Substances Data | 75-45-6(Hazardous Substances Data) |
| Difluorochloromethane Usage And Synthesis |
| Chemical Properties | colourless gas |
| General Description | Difluorochloromethane is a colorless gas with an ethereal odor. Difluorochloromethane is shipped as a liquefied gas under its own vapor pressure. Difluorochloromethane is noncombustible. Difluorochloromethane can asphyxiate by the displacement of air. Contact with the liquid can cause frostbite. Toxic gases can be produced in fires involving Difluorochloromethane. Exposure of the container to prolonged heat or fire may cause Difluorochloromethane to rupture violently and rocket. |
| Air & Water Reactions | The liquefied gas poured into water can be violently explosive. This is due to the phase transition from superheated liquid to vapor. |
| Reactivity Profile | Difluorochloromethane is incompatible with the following: Alkalis, alkaline earth metals (e.g., powdered aluminum, sodium, potassium, zinc) . |
| Health Hazard | Inhalation at greater than 10% concentration in air may cause narcosis. Liquid may cause frostbite. |
| Fire Hazard | Special Hazards of Combustion Products: Decomposition gases are toxic and irritating. |
Company: |
QUZHOU LANDE MATERIAL TRADE CO.,LTD |
Contact: |
Zhangjin |
Phone: |
+86-0570-8883053 |
Mobile phone: |
+8613511405822 |
Fax: |
+86-0570-3800039 |
E-mail: |
sales@nb-skss.com |
Website:
|
http://www.qzldchem.com |
Address: |
No.48, Donggang 1st Road, green industry cluster district, Quzhou City |
Zip Code: |
324000 |