Vonoprazan Fumarate
-
- Category :
Pharmaceuticals and Biochemicals
- CAS NO : 1260141-27-2
- EC NO :
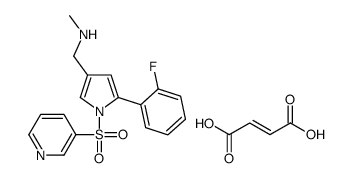
- Molecular Formula : C21H20FN3O6S
- Main Specifications : 99% up
- Synonyms : Vonoprazan fumarate;TAK-438;TAK 438;5-(2-Fluorophenyl)-N-methyl-1-(3-pyridinylsulfonyl)-1H-pyrrole-3-methanamine (2E)-2-butenedioate;Vonaprazan;TAK438;
Package: 25kg/drum
Uses : Corrosive esophagitis (erosiveoesophagitis, EO)
Molecular Structure:
Product description:
Function of Vonoprazan Fumarate
1) Prevention of duodenal ulcer and gastric ulcer recurrence
A randomized, double-blind, multi-center clinical phase 3 trial comparing the effects of Voronazan fumarate (10mgqd and 20mgqd) and lansoprazole (15mgqd) on nonsteroidal anti-inflammatory drug-related peptic ulcer A total of 642 patients had been diagnosed with endoscopic peptic ulcers and needed to take non-steroidal anti-inflammatory drugs. The treatment period was 24 weeks. The primary endpoint was the proportion of duodenal ulcer and gastric ulcer recurrence at 24 weeks.
2) Corrosive esophagitis (erosiveoesophagitis, EO)
In a randomized, double-blind, multi-center, dose-range clinical phase 2 trial, in patients with EO, compared with lansoprazole, Voronazan fumarate showed non-inferiority and was rated in Los Angeles as C/D grade patients showed excellent effects, and oral administration of 20 mg once daily became the clinically recommended dose for the treatment of EO. A randomized, double-blind, multi-center clinical phase 3 trial comparing the efficacy of this product (20mgqd) and lansoprazole (30mgqd) on EO, a total of 409 patients participated in the study.
3) Helicobacter pylori infection (Helicobacterpylori, Hp)
A randomized, double-blind, multi-center clinical phase 3 trial comparing vonolazan fumarate (20mgbid) and lansoprazole (30mgbid), combined with amoxicillin and clarithromycin, to form a triple therapy, The effect of first-line medicine to eradicate Hp was included in a total of 650 Hp-positive patients who had had gastric ulcer or duodenal ulcer. The Hp eradication rates of this product and lansoprazole in the test group were 92.6% and 75.9%, respectively. For patients with clarithromycin resistance, the Hp eradication rates in the two test groups were 82.0% and 40.0%, respectively. Significantly superior to lansoprazole. In this trial, the first 50 patients who failed first-line treatment received second-line treatment with triple therapy of this product, amoxicillin and metronidazole, and the Hp clearance rate was 98%.
