Dantrolene sodium
Inquiry
| Post Date: | Jan 09,2018 |
| Expiry Date: | Jul 08,2018 |
| Detailed Description: |
Cas No. :24868-20-0
Quantity: mass in stockKilograms Specs:Orange powder Price:negotiable USD Kilograms Payment Method: T/T, Western Union,Moneygram and Bitcoin. Chemically it is a hydantoin derivative, but does not exhibit antiepileptic activity like other hydantoin derivates such as phenytoin. The poor water solubility of dantrolene leads to certain difficulties in its use. A more water-soluble analog of dantrolene, azumolene, is under development for similar indications. Azumolene has a bromine residue instead of the nitro group found in dantrolene, and is 30 times more water-soluble. Dantrolene sodium is a postsynaptic muscle relaxant that lessens excitation-contraction coupling in muscle cells. It achieves this by inhibiting Ca2+ ions release from sarcoplasmic reticulum stores by antagonizing ryanodine receptors. It is the primary drug used for the treatment and prevention of malignant hyperthermia, a rare, life-threatening disorder triggered by general anesthesia. It is also used in the management of neuroleptic malignant syndrome, muscle spasticity (e.g. after strokes, in paraplegia, cerebral palsy, or patients with multiple sclerosis), and 2,4-dinitrophenol poisoning, and the related compounds dinoseb and dinoterb. It is marketed by Par Pharmaceuticals LLC as Dantrium (in North America) and by Norgine BV as Dantrium, Dantamacrin, or Dantrolen (in Europe). A hospital is recommended to keep a minimum stock of 36 dantrolene vials totaling 720 mg, sufficient for a 70-kg person. Dantrolene was first described in the scientific literature in 1967, as one of several hydantoin derivatives proposed as a new class of muscle relaxant. Dantrolene underwent extensive further development, and its action on skeletal muscle was described in detail in 1973. Dantrolene was widely used in the management of spasticity before its efficacy in treating malignant hyperthermia was discovered by South African anesthesiologist Gaisford Harrison and reported in a landmark 1975 article published in the British Journal of Anaesthesia. Harrison experimentally induced malignant hyperthermia with halothane anesthesia in genetically susceptible pigs, and obtained an 87.5% survival rate, where seven of his eight experiments survived after intravenous administration of dantrolene. The efficacy of dantrolene in humans was later confirmed in a large, multicenter study published in 1982, and confirmed epidemiologically in 1993. Before dantrolene, the only available treatment for malignant hyperthermia was procaine, which was associated with a 60% mortality rate in animal models. Oral dantrolene cannot be used by: people with a pre-existing liver disease people with compromised lung function people with severe cardiovascular impairment people with a known hypersensitivity to dantrolene pediatric patients under five years of age people who need good muscular balance or strength to maintain an upright position, motoric function, or proper neuromuscular balance If the indication is a medical emergency, such as malignant hyperthermia, the only significant contraindication is hypersensitivity. If needed in pregnancy, adequate human studies are lacking, therefore the drug should be given in pregnant women only if clearly indicated. It may cause hypotonia in the newborn if given closely before delivery. Dantrolene should not be given to breastfeeding mothers. If a treatment is necessary, breastfeeding should be terminated. Central nervous system side effects are quite frequently noted and encompass speech and visual disturbances, mental depression and confusion, hallucinations, headache, insomnia and exacerbation or precipitation of seizures, and increased nervousness. Infrequent cases of respiratory depression or a feeling of suffocation have been observed. Dantrolene often causes sedation severe enough to incapacitate the patient to drive or operate machinery. Gastrointestinal effects include bad taste, decreased appetite, nausea, vomiting, abdominal cramps, and diarrhea. Liver side effects may be seen either as asymptomatic elevation of liver enzymes and/or bilirubin or, most severe, as fatal and nonfatal liver inflammation. The risk of liver inflammation is associated with the duration of treatment and the daily dose. In patients treated for hyperthermia, no liver toxicity has been observed so far. Pleural effusion with inflammation of the fibrous sac around the heart (oral treatment only), rare cases of bone marrow damage, diffuse muscle pains, backache, dermatologic reactions, transient cardiovascular reactions, and crystals in the urine have additionally been seen. Muscle weakness may persist for several days following treatment. |
| CAS Registry Number: | 24868-20-0 |
| Synonyms: | ;Dantrolene, Sodium Salt;1-[[5-(p-Nitrophenyl)furfurylidene]amino]-hydantoin sodium salt; 1-[5-(-4-Nitrophenyl)furfurylideneamino]imidazolidine-2,4-dione sodium;sodium 1-({(1E)-[5-(4-nitrophenyl)furan-2-yl]methylidene}amino)-4-oxo-4,5-dihydro-1H-imidazol-2-olate;sodium 3-({[5-(4-nitrophenyl)furan-2-yl]methylidene}amino)-2,5-dioxoimidazolidin-1-ide;2,4-imidazolidinedione, 1-[[(1E)-[5-(4-nitrophenyl)-2-furanyl]methylene]amino]-, sodium salt;Dantrolene sodium salt;Dantrolene Na; |
| Molecular Formula: | C14H10N4NaO5 |
| Molecular Weight: | 337.2428 |
| Molecular Structure: | 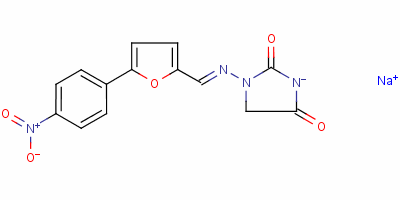
|
| Safety Description: | S22:; S24/25:; |
| Company: | Hengyang Desen Biotechnology Co., Ltd. [ China ] |
| Contact: | ava |
| Tel: | 86-18908446905 |
| Fax: | |
| Email: | ava@ycgmp.com |
-
Disclaimer statement:The information and data included above have been realized by the enterprises and compiled by the staff, and are subject to change without notice to you. The Chemnet makes no warranties or representations whatsoever regarding the facticity, accuracy and validity of such information and data. In order to ensure your interest, we suggest you chose the products posted by our gold suppliers or VIP members.