Lubiprostone
-
Post Date:
Feb 04,2015
-
Expiry Date:
Feb 04,2016
-
Detailed Description:
Cas No. :136790-76-6
Product name: Lubiprostone
Description: Lubiprostone (rINN, marketed under the trade name Amitiza) is a medication used in the management of chronic idiopathic constipation, predominantly constipation irritable bowel syndrome in WOMEN, and opioid-induced constipation . It was initially approved by the U.S. Food and Drug Administration (FDA) on 31 January 2006.
Lubiprostone is used for the treatment of chronic constipation of unknown cause in adults, as well as irritable bowel syndrome associated with constipation in women.
As of 20 July 2006, Lubiprostone has not been studied in children. There is current research underway to determine the efficacy in postoperative bowel dysfunction.
-
CAS Registry Number:
136790-76-6
-
Synonyms:
;7-[(1r,4r,6r,9r)-4-(1,1-difluoropentyl)-4-hydroxy-8-oxo-5-oxabicyclo[4.3.0]non-9-yl]heptanoic acid;(11a)-16,16-Difluoro-11-hydroxy-9,15-dioxo-prostan-1-oic Acid;(11alpha,15R)-16,16-difluoro-15-hydroxy-9-oxo-11,15-epoxyprostan-1-oic acid;(8beta,11alpha,15R)-16,16-difluoro-15-hydroxy-9-oxo-11,15-epoxyprostan-1-oic acid;(2R,4aR,5R,7aR)-2-(1,1-Difluoropentyl)-2-hydroxy-6-oxo-3,4,4a,5,7,7a-hexahydrocyclopenta[b]pyran-5-heptanoic acid;7-((2R,4aR,5R,7aR)-2-(1,1-Difluoropentyl)-2-hydroxy-6-oxooctahydrocyclopenta[b]pyran-5-yl)heptanoic acid;
-
Molecular Formula:
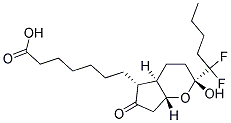
C20H32F2O5
-
Molecular Weight:
390.4619
-
Molecular Structure:
-
Company:
AUSMAUCO BIOTECH CO., LIMITED
[ China ]
-
Contact:
john
-
Tel:
+8618060810639
-
Fax:
-
Email:
plantsale@aus-bio.com
Inquiry
